What Is Semaglutide?
Semaglutide is a GLP-1 receptor agonist used for Type 2 diabetes (brand: Ozempic) and chronic weight management (brand: Wegovy). It mimics the incretin hormone GLP-1, slowing gastric emptying, reducing appetite, and improving glycemic control. Its long half-life of approximately 168 hours (7 days) allows once-weekly dosing.
FDA-Approved Dose Escalation: Wegovy
The Wegovy schedule is designed to minimize GI side effects by slowly increasing the dose over 16 weeks:
• Weeks 1-4: 0.25 mg/week (initiation dose)
• Weeks 5-8: 0.5 mg/week
• Weeks 9-12: 1.0 mg/week
• Weeks 13-16: 1.7 mg/week
• Week 17+: 2.4 mg/week (maintenance dose)
Do not skip escalation steps. If you experience persistent nausea, remain on your current dose for an additional 4 weeks before escalating.
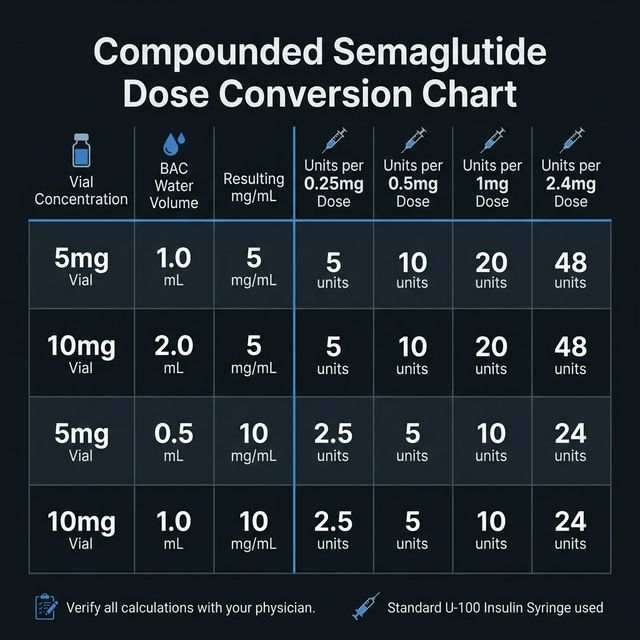
How to Calculate Syringe Units for Compounded Semaglutide
Compounded semaglutide typically comes as a lyophilized powder. After reconstitution, use this formula:
Example: You have a 5 mg vial reconstituted with 2.5 mL of BAC water. Concentration = 2 mg/mL. For a 0.25 mg dose using a 100-unit syringe: Units = 0.25 ÷ 2 × 100 = 12.5 units (round to 13 units).
Injection Technique
• Inject subcutaneously in the abdomen, thigh, or upper arm
• Rotate injection sites weekly to prevent lipodystrophy
• Inject at the same time each week (± 2 days if you miss a dose)
• Store reconstituted solution at 2-8°C
Side Effect Management
The most common side effects are gastrointestinal: nausea (44%), diarrhea (30%), vomiting (24%). These typically improve after 4-8 weeks as your body adjusts.
Mitigation strategies: eat smaller, more frequent meals; avoid high-fat or spicy foods; stay hydrated; and slow down your dose escalation if symptoms are severe.
Clinical Efficacy
In the STEP 1 trial (n=1,961), patients on Wegovy 2.4 mg lost an average of 14.9% body weight vs. 2.4% with placebo over 68 weeks. Approximately 86% of participants achieved ≥5% weight loss.
Source: Wilding JPH, et al. N Engl J Med. 2021;384:989-1002. DOI: 10.1056/NEJMoa2032183