⚕️ Medical Disclaimer
**⚕️ Medical Disclaimer:** This article is for educational and informational purposes only. It does not constitute medical advice. Consult a qualified healthcare provider before using any medication.
What Is CagriSema?
CagriSema is Novo Nordisk's fixed-ratio combination of **semaglutide** (a GLP-1 receptor agonist) and **cagrilintide** (a long-acting amylin analog) in a single weekly subcutaneous injection. The REDEFINE 1 pivotal trial demonstrated **22.7% mean weight loss at 68 weeks** — the largest weight reduction ever reported for a single medication in an obesity trial (Lau et al., 2024, NEJM). FDA filing (NDA) was submitted in late 2024 with potential approval expected in 2025-2026.
*Last updated: March 2026*
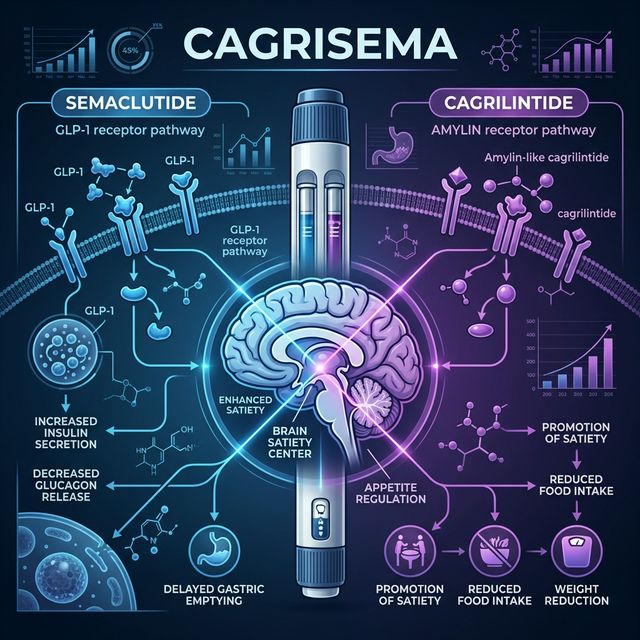
How Does CagriSema Work? Dual Mechanism Explained
**Semaglutide component:** GLP-1 receptor agonist that suppresses appetite centrally (hypothalamic POMC/CART activation, NPY/AgRP inhibition), slows gastric emptying, and enhances glucose-dependent insulin secretion. This is the same molecule in Ozempic and Wegovy.
**Cagrilintide component:** Long-acting acylated analog of amylin — a peptide co-secreted with insulin from pancreatic β-cells. Amylin suppresses glucagon secretion, slows gastric emptying through a different pathway than GLP-1, and activates the area postrema for satiety signaling. By combining two distinct satiety pathways, CagriSema achieves synergistic appetite suppression.
For semaglutide dosing on its own, see our semaglutide dose calculator and GLP-1 escalation guide.
CagriSema Clinical Results: REDEFINE Trials
| Trial | Population | Duration | Weight Loss | Comparison |
|---|---|---|---|---|
| REDEFINE 1 | Adults with obesity (BMI ≥30) | 68 weeks | -22.7% | Placebo: -2.3% |
| REDEFINE 2 | T2D + obesity | 68 weeks | -15.7% | Semaglutide alone: -12.2% |
| REDEFINE 3 | Sequential after semaglutide | 68 weeks | Additional -8% beyond sema | — |
Key finding from REDEFINE 2: CagriSema produced 3.5% MORE weight loss than semaglutide alone — demonstrating that the cagrilintide component adds meaningful incremental benefit through the amylin pathway.
CagriSema vs Tirzepatide vs Retatrutide: Next-Gen Comparison
| Feature | CagriSema | Tirzepatide (Mounjaro) | Retatrutide |
|---|---|---|---|
| Mechanism | GLP-1 + Amylin | GLP-1 + GIP | GLP-1 + GIP + Glucagon |
| Max Weight Loss | ~23% (68 wk) | ~22.5% (72 wk) | ~24% (48 wk, Phase 2) |
| Route | SubQ weekly | SubQ weekly | SubQ weekly |
| FDA Status | NDA filed (2024) | Approved (2023) | Phase 3 |
| Manufacturer | Novo Nordisk | Eli Lilly | Eli Lilly |
| Muscle Preservation | Under study | Better than sema | Under study |
This is the new generation of obesity medicine. For current GLP-1 scheduling, see the GLP-1 Scheduler Calculator.
Frequently Asked Questions
**When will CagriSema be available?** FDA NDA filed late 2024. Potential approval mid-2025 to early 2026. Brand name and pricing not yet finalized.
**Can I create my own CagriSema by combining semaglutide + cagrilintide?** This is not recommended. The fixed-ratio combination in CagriSema was specifically designed for optimal pharmacokinetics. Mixing separate vials introduces dosing inconsistencies.
**Is CagriSema better than tirzepatide?** Head-to-head trials have not been completed. Both achieve ~22-23% weight loss through different dual-mechanism approaches (GLP-1+amylin vs GLP-1+GIP).
**What are the side effects?** Similar to semaglutide: nausea, vomiting, diarrhea, constipation. The amylin component may add injection site reactions. Cardiovascular outcomes trials are ongoing.
**Will CagriSema be covered by insurance?** Pricing and coverage will be determined upon FDA approval. Expected to be in the same cost range as current branded GLP-1 agonists (~$1,000-1,500/month without insurance).