⚕️ Medical Disclaimer
**⚕️ Medical Disclaimer:** This article is for educational and informational purposes only. It does not constitute medical advice. Consult a qualified healthcare provider before using any peptide.
The Oral Semaglutide Revolution: From 14 mg to 50 mg
Oral semaglutide (Rybelsus) launched in 2019 at doses of 3, 7, and 14 mg daily for type 2 diabetes. The problem: even at 14 mg, its weight loss efficacy was about half of injectable semaglutide 2.4 mg (Wegovy). The peptide community and Novo Nordisk both knew that the oral dose needed to go much higher to compete with injections.
*Last updated: March 2026*
Enter the OASIS clinical trial program, which tested oral semaglutide at 25 mg and 50 mg daily — doses that approached and, in some endpoints, matched injectable Wegovy's weight loss performance. This represents a potential paradigm shift: eliminating the need for weekly injections while maintaining equivalent efficacy.
This guide covers the clinical data, the pharmacokinetic challenges of oral peptide delivery, comparative efficacy with injectable semaglutide, and what these developments mean for the broader GLP-1 landscape. For current injectable semaglutide dosing, use our Semaglutide Dose Calculator and GLP-1 Dose Escalation Guide.
Why Does Oral Semaglutide Need Such High Doses?
**Oral bioavailability of semaglutide is approximately 0.4-1% — meaning 99+ percent of the ingested dose is destroyed by stomach acid and proteolytic enzymes before absorption.** This is the fundamental challenge of oral peptide delivery.
Rybelsus uses SNAC (sodium N-[8-(2-hydroxybenzoyl)amino]caprylate) as an absorption enhancer. SNAC creates a local pH increase around the tablet in the stomach lining, temporarily protecting semaglutide from degradation and facilitating transcellular absorption. Even with this technology, absorption is highly variable and requires strict fasting conditions:
• Take on an empty stomach with no more than 120 mL (4 oz) of plain water
• Wait at least 30 minutes before eating, drinking, or taking other medications
• Take at the same time each day (morning recommended)
Violation of any of these conditions reduces absorption dramatically — by up to 70-80% in some studies. This is why oral semaglutide requires doses 20-50x higher than injectable to achieve similar plasma levels.
The math: injectable Wegovy at 2.4 mg weekly delivers approximately 0.34 mg/day of active semaglutide. Oral semaglutide at 50 mg daily delivers approximately 0.25-0.5 mg/day after accounting for ~1% bioavailability — roughly equivalent systemic exposure.
OASIS Clinical Trial Results: 25 mg and 50 mg Data
**The OASIS program is Novo Nordisk's Phase 3 trial series evaluating high-dose oral semaglutide for obesity:**
**OASIS 1 (published 2023):** 667 adults with obesity, randomized to oral semaglutide 50 mg daily vs placebo for 68 weeks (Knop et al., 2023, PMID: 37385280).
• Mean weight loss: **-15.1%** (50 mg) vs -2.4% (placebo)
• ≥5% weight loss: 85% of participants
• ≥10% weight loss: 69% of participants
• ≥20% weight loss: 34% of participants
**OASIS 4:** Oral semaglutide 50 mg vs placebo in adults with obesity and knee osteoarthritis. Demonstrated significant weight loss and improvement in knee pain scores. GI side effects were consistent with the GLP-1 class.
**Context:** Injectable Wegovy 2.4 mg achieves approximately 15-17% weight loss in pivotal trials (STEP program). The OASIS 1 result of 15.1% with oral 50 mg is in the same range — suggesting approximate clinical equivalence for the first time.
| Formulation | Dose | Route | Weight Loss (%) | Key Trial | Convenience Factor |
|---|---|---|---|---|---|
| Rybelsus (current) | 14 mg daily | Oral | ~5-7% | PIONEER | Low (strict fasting, low efficacy) |
| Oral semaglutide (high-dose) | 25 mg daily | Oral | ~10-13% | OASIS 2 | Moderate (fasting required) |
| Oral semaglutide (high-dose) | 50 mg daily | Oral | ~15.1% | OASIS 1 | Moderate (fasting required) |
| Wegovy (injectable) | 2.4 mg weekly | SubQ injection | ~15-17% | STEP | High (weekly injection, no fasting) |
| Tirzepatide (Zepbound) | 15 mg weekly | SubQ injection | ~20-22.5% | SURMOUNT | High (weekly injection) |
Oral vs Injectable Semaglutide: Which Is Better?
**Both formulations deliver the same molecule — semaglutide. The differences are practical, not pharmacological:**
**Arguments for oral (25-50 mg):**
• No needles — eliminates injection anxiety and injection site reactions
• No cold chain requirements (tablets are temperature-stable)
• No reconstitution math or syringe draw calculations
• Travel-friendly — no TSA concerns, no cooler bags (see our travel guide)
• Daily dosing may provide more stable plasma levels than weekly injection peaks/troughs
**Arguments for injectable (Wegovy 2.4 mg):**
• Once-weekly dosing (7x fewer dose events than daily oral)
• No fasting requirement — inject any time
• More predictable absorption (not affected by food, water volume, or timing)
• Shorter titration period to maximum dose
• Auto-injector pens are simple and painless (see our injection guide)
• Currently better insurance coverage (Wegovy has been on-market longer)
**The likely answer:** Patient preference will drive choice once both high-dose oral and injectable are approved for obesity. Needle-averse patients benefit from oral. Patients who struggle with daily fasting compliance benefit from weekly injectable. Discuss with your prescriber.
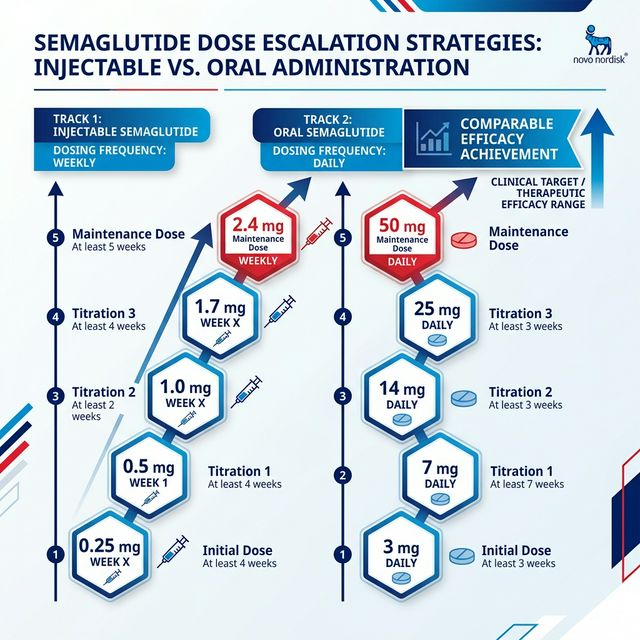
Dose Escalation for High-Dose Oral Semaglutide
**Like injectable semaglutide, oral semaglutide requires gradual dose escalation to minimize GI side effects:**
| Week | Oral Dose (Daily) | Purpose |
|---|---|---|
| Weeks 1-4 | 3 mg | GI tolerance initiation |
| Weeks 5-8 | 7 mg | Intermediate escalation |
| Weeks 9-12 | 14 mg | Standard approved dose (Rybelsus) |
| Weeks 13-16 | 25 mg | High-dose therapeutic range |
| Weeks 17+ | 50 mg | Maximum studied dose |
Each dose increase carries a 2-4 week GI adaptation period (nausea, occasional vomiting, decreased appetite). The 14→25 mg and 25→50 mg steps seem to produce the largest GI burden in clinical trials. Do not rush escalation — GI intolerance is the primary reason patients discontinue GLP-1 therapy.
For injectable dose escalation guidance, see our GLP-1 Dose Escalation Guide. For dose calculations with injectable semaglutide, use our Semaglutide Dose Calculator.
Side Effects at High Doses
**The side effect profile of high-dose oral semaglutide is consistent with the GLP-1 class but intensified in frequency and severity at 25-50 mg:**
• **Nausea:** 30-40% at 50 mg (vs ~15% at 14 mg). Most resolves within 4-8 weeks of each dose escalation.
• **Vomiting:** ~10-15% at 50 mg. Can be significant enough to cause dehydration — maintain fluid intake.
• **Diarrhea/constipation:** 10-20% at 50 mg. Fiber supplementation and hydration help.
• **Decreased appetite:** Expected — this is the therapeutic mechanism. Monitor for excessive restriction.
• **Pancreatitis risk:** Rare but serious. Report severe upper abdominal pain immediately. Family history of MEN-2 syndrome or personal history of medullary thyroid carcinoma are absolute contraindications.
• **Gallbladder disease:** Rapid weight loss increases gallstone risk. Consider prophylactic ursodeoxycholic acid if losing >1.5% body weight per week.
• **Lean mass loss:** 30-40% of weight lost on any GLP-1 therapy is lean mass unless counteracted with resistance training and high protein intake (1.6-2.2 g/kg/day). Non-negotiable.
Monitor with regular blood work: lipase/amylase (pancreatitis screening), liver enzymes (hepatobiliary monitoring), HbA1c, and kidney function.
What This Means for the Future of GLP-1 Therapy
**High-dose oral semaglutide represents the next competitive frontier in the GLP-1 market:**
• **Novo Nordisk** is filing for approval of oral semaglutide 25 mg and 50 mg for obesity, potentially launching as "oral Wegovy" in 2026-2027.
• **Eli Lilly** is developing its own oral GLP-1 compounds, including orforglipron (non-peptide oral GLP-1R agonist) — see our orforglipron guide.
• **Amgen** has an oral obesity drug (MariTide, non-daily oral anti-GIPR antibody + GLP-1 agonist) in development.
• **The patient experience question:** Will patients prefer a daily tablet with fasting requirements or a weekly injection with no food restrictions? Market research suggests a near-even split, with younger patients and needle-phobic patients favoring oral.
• **Cost implications:** Oral formulations are typically easier to manufacture at scale than pre-filled injection pens. This could reduce production costs, though pricing decisions are ultimately market-driven.
• **Compounding impact:** Oral semaglutide cannot be easily compounded — the SNAC absorption enhancer is patent-protected. Injectable semaglutide compounding will remain the primary alternative for price-sensitive patients. For survodutide and the broader dual-agonist pipeline, see our survodutide guide.
Frequently Asked Questions
**Is oral semaglutide 50 mg available now?** As of March 2026, oral semaglutide 50 mg is being studied in Phase 3 trials (OASIS program) but is not yet FDA-approved for obesity. Current approved oral semaglutide (Rybelsus) is available at 3, 7, and 14 mg for type 2 diabetes only.
**Can I just take more Rybelsus tablets to get a 50 mg dose?** This is strongly discouraged. The SNAC absorption enhancer works best with a single tablet — splitting or stacking multiple tablets changes the gastric pH dynamics and absorption kinetics unpredictably. Additionally, exceeding the approved dose without medical supervision is risky.
**Will oral semaglutide replace Wegovy injections?** Probably not replace, but offer an alternative. Many patients prefer the simplicity of a weekly injection over daily fasting-dependent oral dosing. Both formulations will likely coexist for different patient preferences.
**How does oral semaglutide compare to orforglipron?** Both are oral GLP-1R agonists. Semaglutide is a peptide requiring the SNAC absorption enhancer and fasting conditions. Orforglipron is a small molecule that does not require fasting and has good oral bioavailability naturally. Orforglipron may offer superior convenience but semaglutide has a much larger safety dataset. See our orforglipron guide.
**What BMI is needed to qualify for semaglutide?** Generally, a BMI ≥30 (or ≥27 with a weight-related comorbidity) qualifies for prescription semaglutide for weight management. Our colleagues at BMI Calc Now offer a comprehensive BMI calculator if you need to check your current BMI.
Final Word
**⚕️ Medical Disclaimer:** This article is for educational and informational purposes only. It does not constitute medical advice. Consult a qualified healthcare provider before using any peptide.