GLP-1 Dose Escalation Guide: Semaglutide, Tirzepatide & Retatrutide
Comprehensive guide to GLP-1 agonist dosing schedules for semaglutide (Wegovy/Ozempic), tirzepatide (Mounjaro/Zepbound), and retatrutide with syringe unit calculations and clinical data.
Key Takeaways
- ✓GLP-1 agonists require 4-20 weeks of gradual dose escalation to minimize GI side effects
- ✓Semaglutide (Wegovy) escalates from 0.25 mg to 2.4 mg over 16+ weeks
- ✓Tirzepatide (Mounjaro/Zepbound) achieves the greatest weight loss at 22.5% in clinical trials
- ✓Units = Dose (mg) ÷ Concentration (mg/mL) × 100 — use our calculator for instant results
- ✓Never skip escalation steps — each 4-week period lets your body adapt to slower gastric emptying
⚕️ Medical Disclaimer: This guide is for educational purposes only. Consult a licensed healthcare provider before starting, stopping, or changing any GLP-1 agonist.
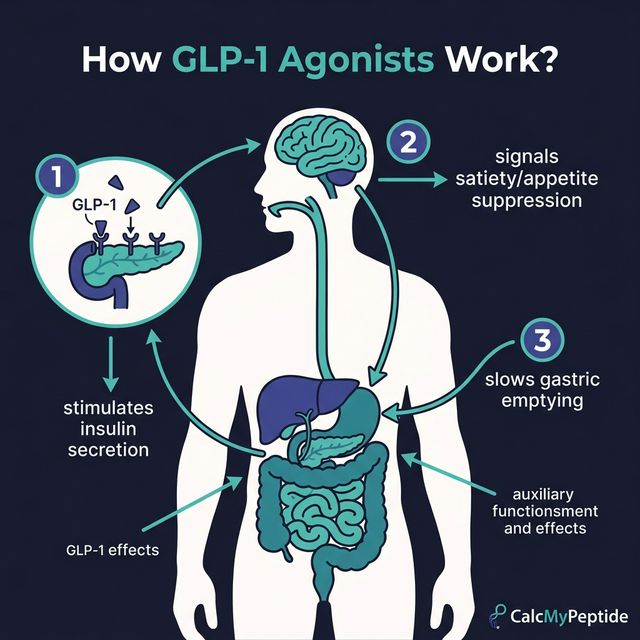
1. What Are GLP-1 Receptor Agonists?
GLP-1 receptor agonists are a class of medications that mimic glucagon-like peptide-1 (GLP-1), a naturally occurring incretin hormone. When food enters the digestive system, the gut releases GLP-1, which signals the pancreas to produce insulin, suppresses glucagon secretion, slows gastric emptying, and communicates satiety to the brain.
Synthetic GLP-1 agonists amplify these effects far beyond what natural GLP-1 can achieve. They have been FDA-approved for type 2 diabetes management since 2005 (exenatide/Byetta) and for chronic weight management since 2014 (liraglutide/Saxenda), with dramatically more effective agents introduced since 2021.
GLP-1 Mono
Semaglutide (Ozempic, Wegovy)
Single incretin — targets GLP-1 receptor only
GIP/GLP-1 Dual
Tirzepatide (Mounjaro, Zepbound)
Dual incretin — activates both GIP and GLP-1 receptors
GIP/GLP-1/Glucagon Triple
Retatrutide (Phase III)
Triple agonist — adds glucagon receptor for increased energy expenditure
2. Why Dose Escalation Is Required
GLP-1 agonists require gradual dose escalation over 4-20 weeks to minimize gastrointestinal side effects — primarily nausea, vomiting, diarrhea, and constipation. Starting at the full therapeutic dose often causes intolerable GI distress and leads to treatment discontinuation.
The reason: GLP-1 receptor activation significantly slows gastric emptying (the rate food leaves the stomach). At low doses, the body gradually adapts to this slower transit time. Jumping directly to a high dose overwhelms the GI system before adaptation can occur.
Key principle: Each dose step is maintained for 4 weeks minimum before escalating. Some clinicians extend to 8 weeks if side effects persist. Escalation should not proceed until current-dose side effects are manageable.
Dose escalation also allows clinicians to find the minimum effective dose for each patient. Not everyone needs the maximum dose — many patients achieve satisfactory results at intermediate dose steps, with fewer side effects and lower cost.
3. Semaglutide: Wegovy & Ozempic Schedules
Semaglutide has a ~7-day half-life, making once-weekly injection convenient and effective. Two FDA-approved formulations exist with different indication-specific dose targets:
Wegovy (Weight Management)
Weeks 1-4 → 0.25 mg
Weeks 5-8 → 0.5 mg
Weeks 9-12 → 1.0 mg
Weeks 13-16 → 1.7 mg
Week 17+ → 2.4 mg (maintenance)
Source: FDA Label (Wegovy)
Ozempic (Type 2 Diabetes)
Weeks 1-4 → 0.25 mg
Weeks 5-8 → 0.5 mg
Week 9+ → 1.0 mg (up to 2.0 mg)
Source: FDA Label (Ozempic)
The weight management dose target (2.4 mg) is significantly higher than the diabetes dose (1.0-2.0 mg), reflecting the STEP clinical trial findings that higher doses produce greater weight loss. Average weight loss in the STEP 1 trial was 14.9% at the 2.4 mg dose over 68 weeks.
4. Tirzepatide: Mounjaro & Zepbound Schedules
Tirzepatide is a dual GIP/GLP-1 receptor agonist with a ~5-day half-life. By activating both incretin receptors, it achieves greater glycemic control and weight loss than GLP-1-only agents in head-to-head trials.
Mounjaro / Zepbound Schedule
Weeks 1-4 → 2.5 mg
Weeks 5-8 → 5.0 mg
Weeks 9-12 → 7.5 mg (optional step)
Weeks 13-16 → 10.0 mg
Weeks 17-20 → 12.5 mg (optional step)
Week 21+ → 15.0 mg (maintenance)
Source: FDA Label (Mounjaro/Zepbound)
The SURMOUNT-1 trial showed tirzepatide at the maximum dose achieved 22.5% mean weight loss over 72 weeks — the largest weight reduction seen in a Phase III obesity trial at the time of publication.
5. Retatrutide: The Triple Agonist
Retatrutide (LY3437943) is an investigational triple agonist activating GLP-1, GIP, and glucagon receptors simultaneously. The glucagon receptor activation adds thermogenesis (increased energy expenditure) on top of the appetite suppression and insulin secretion. Half-life: ~6 days.
Retatrutide Phase II Schedule
Weeks 1-4 → 1.0 mg
Weeks 5-8 → 2.0 mg
Weeks 9-12 → 4.0 mg
Weeks 13-16 → 8.0 mg
Week 17+ → 12.0 mg (maintenance)
Source: PMID: 37351564 (Phase II)
Phase II results showed up to 24.2% mean weight loss at the 12 mg dose over 48 weeks. Retatrutide is currently in Phase III trials (TRIUMPH program) with expected results in 2025-2026.
6. Oral Semaglutide (Rybelsus)
Rybelsus is the oral formulation of semaglutide, FDA-approved for type 2 diabetes. It uses SNAC (sodium N-[8-(2-hydroxybenzoyl)amino]caprylate) as an absorption enhancer to allow the peptide to survive stomach acid and be absorbed through the gastric lining.
Rybelsus Dose Escalation
Days 1-30 → 3 mg daily (starter dose, not therapeutic)
Days 31-60 → 7 mg daily
Day 61+ → 14 mg daily (maintenance)
Source: FDA Label (Rybelsus)
⚠️ Important: Oral semaglutide must be taken on an empty stomach with no more than 4 oz (120 mL) of plain water. Wait at least 30 minutes before eating, drinking, or taking other oral medications. Food significantly reduces absorption.
Key difference: The oral bioavailability of semaglutide is only about 1% compared to subcutaneous injection, which is why oral doses (3-14 mg/day) are much higher than injectable doses (0.25-2.4 mg/week). The clinical efficacy for weight loss is somewhat lower than injectable semaglutide, though oral semaglutide 50 mg (under investigation) may close that gap.
7. Reconstituting Compounded GLP-1 Peptides
Compounded semaglutide and tirzepatide typically come as lyophilized powder in vials of 3 mg, 5 mg, or 10 mg. Unlike brand-name auto-injectors (which are pre-mixed), compounded versions require reconstitution with bacteriostatic water.
The reconstitution math is identical to any other peptide — concentration equals vial size divided by water volume. Use our Reconstitution Calculator for instant results.
| Vial | Water | Conc. | 0.25 mg dose | 1.0 mg dose | 2.4 mg dose |
|---|---|---|---|---|---|
| 5 mg | 2 mL | 2.5 mg/mL | 10 units | 40 units | 96 units |
| 5 mg | 1 mL | 5 mg/mL | 5 units | 20 units | 48 units |
| 3 mg | 1 mL | 3 mg/mL | 8.3 units | 33.3 units | 80 units |
| 10 mg | 2 mL | 5 mg/mL | 5 units | 20 units | 48 units |
8. Calculating Syringe Units for Each Dose Step
Units = Dose (mg) ÷ Concentration (mg/mL) × 100
Example for Wegovy schedule with 5 mg / 2 mL reconstitution (2.5 mg/mL):
| Phase | Dose | Calculation | Draw | Syringe |
|---|---|---|---|---|
| Month 1 | 0.25 mg | 0.25 ÷ 2.5 × 100 | 10 units | 30u syringe |
| Month 2 | 0.5 mg | 0.5 ÷ 2.5 × 100 | 20 units | 30u syringe |
| Month 3 | 1.0 mg | 1.0 ÷ 2.5 × 100 | 40 units | 50u syringe |
| Month 4 | 1.7 mg | 1.7 ÷ 2.5 × 100 | 68 units | 100u syringe |
| Maintenance | 2.4 mg | 2.4 ÷ 2.5 × 100 | 96 units | 100u syringe |
9. Managing Side Effects During Escalation
The most common side effects of GLP-1 agonists are gastrointestinal. They typically peak during the first 1-2 weeks of each new dose level and diminish with continued use.
Nausea
Eat smaller meals, avoid greasy/spicy food, stay hydrated. Usually resolves within 1-2 weeks.
Constipation
Increase fiber intake, drink more water, consider osmotic laxatives (PEG 3350).
Injection site reactions
Rotate injection sites between abdomen, thigh, and upper arm. Apply ice before injection.
Fatigue
Common during first week of escalation. Ensure adequate protein intake (≥1 g/kg) and hydration.
Acid reflux / GERD
Elevated GLP-1 slows gastric emptying, which can worsen reflux. Sleep with upper body elevated; avoid eating 3+ hours before bed.
Hair thinning
Reported in some patients during rapid weight loss. Often related to caloric/protein deficit rather than the drug itself. Ensure ≥1.2 g protein/kg daily.
10. Drug Interactions & Contraindications
GLP-1 agonists slow gastric emptying, which can affect the absorption timing of oral medications taken at the same time. Key interactions to be aware of:
Oral contraceptives
Delayed absorption may reduce efficacy. Take 1 hour before or wait until GLP-1 side effects stabilize.
Sulfonylureas / Insulin
Increased hypoglycemia risk. Dose reduction of sulfonylurea or insulin is often needed when starting GLP-1 therapy.
Levothyroxine
Take on empty stomach 30-60 min before any food or GLP-1 injection. Monitor TSH levels.
Warfarin / Anticoagulants
Monitor INR more frequently during dose escalation as absorption changes may affect warfarin blood levels.
⛔ Contraindications: GLP-1 agonists are contraindicated in patients with a personal or family history of medullary thyroid carcinoma (MTC) or Multiple Endocrine Neoplasia type 2 (MEN2). They should not be used with other GLP-1 receptor agonists. Do not use in pregnancy.
11. Half-Life Comparison Table
| Peptide | Half-Life | Type | Max Dose | Route |
|---|---|---|---|---|
| Semaglutide (injectable) | ~7 days | GLP-1 mono | 2.4 mg/week | SubQ |
| Semaglutide (oral) | ~7 days | GLP-1 mono | 14 mg/day | Oral |
| Tirzepatide | ~5 days | GIP/GLP-1 dual | 15 mg/week | SubQ |
| Retatrutide | ~6 days | GIP/GLP-1/GCG triple | 12 mg/week | SubQ |
| Liraglutide | ~13 hours | GLP-1 mono | 3.0 mg/day | SubQ |
| Exenatide ER | ~2 hours (ER: days) | GLP-1 mono | 2 mg/week | SubQ |
12. Clinical Weight Loss Data
| Trial | Drug | Dose | Duration | Weight Loss |
|---|---|---|---|---|
| STEP 1 | Semaglutide | 2.4 mg/wk | 68 weeks | -14.9% |
| SURMOUNT-1 | Tirzepatide | 15 mg/wk | 72 weeks | -22.5% |
| Phase II | Retatrutide | 12 mg/wk | 48 weeks | -24.2% |
| SCALE | Liraglutide | 3.0 mg/day | 56 weeks | -8.0% |
| OASIS 1 | Oral Semaglutide | 50 mg/day | 68 weeks | -15.1% |
All values are mean placebo-adjusted body weight reduction. Sources: PMID: 33567185, PMID: 35658024, PMID: 37351564.
13. Storage Guidelines
Brand-Name Pens (Wegovy, Mounjaro)
- • Refrigerate at 2-8°C before first use
- • After first use: room temp for up to 56 days (Mounjaro) or 28 days (Wegovy)
- • Protect from light and heat (>86°F/30°C)
- • Do not freeze after reconstitution
Compounded Vials
- • Unreconstituted: freezer (-20°C) for long-term, fridge for 6+ months
- • Reconstituted: always refrigerate at 2-8°C
- • Use within 28 days of adding BAC water
- • Discard if solution is cloudy or discolored
14. Frequently Asked Questions
Can I skip dose escalation steps?
What if I miss a dose during escalation?
Is semaglutide or tirzepatide more effective?
Can I use the same vial across escalation steps?
When will retatrutide be FDA-approved?
Can I take oral semaglutide (Rybelsus) for weight loss?
How much does compounded semaglutide cost vs brand?
Generate Your Personal Schedule
Enter your start date and medication to get a week-by-week escalation plan with syringe units.
Open GLP-1 SchedulerMedical disclaimer: This guide is for educational purposes only. Consult a licensed healthcare provider before starting any GLP-1 agonist. Contact us at contact@calcmypeptide.com.
📖 References
- Wilding JPH, et al. “Once-weekly semaglutide in adults with overweight or obesity (STEP 1).” N Engl J Med (2021). PMID: 33567185
- Jastreboff AM, et al. “Tirzepatide once weekly for the treatment of obesity (SURMOUNT-1).” N Engl J Med (2022). PMID: 35658024
🧪 Try the Calculator
Put this knowledge into practice — calculate your exact concentration and syringe units for free.
Open Reconstitution Calculator →