The Most Important Peptide Nobody Talks About
Thymosin alpha-1 (Tα1) is arguably the most clinically validated peptide in use today — and yet it receives a fraction of the attention that BPC-157, semaglutide, or even MK-677 commands. The reason is context: Tα1 is an immune modulator, not a body composition or aesthetics compound, so it does not appeal to the social media peptide audience. But its clinical evidence base dwarfs most popular peptides.
The numbers: Tα1 has been studied in over 100 clinical trials. It is approved in 35+ countries (as Zadaxin) for hepatitis B, hepatitis C, and as an immune adjuvant in cancer therapy. It has been used in over 25,000 patients in clinical settings. The FDA has granted it orphan drug designation for hepatocellular carcinoma and for the treatment of DiGeorge syndrome.
What Tα1 does: it is a 28-amino-acid peptide naturally produced by the thymus gland that orchestrates immune system maturation, regulation, and response. It is the conductor of the immune orchestra — it does not simply stimulate or suppress the immune system. It modulates it toward appropriate, balanced immune function.
Mechanism: Immune Modulation, Not Stimulation
The distinction between immune stimulation and immune modulation is critical. An immune stimulant makes the immune system more active globally — this can be beneficial when fighting an infection, but harmful in autoimmune conditions where the immune system is already overactive. Tα1 is an immune modulator: it amplifies appropriate immune responses while dampening inappropriate ones.
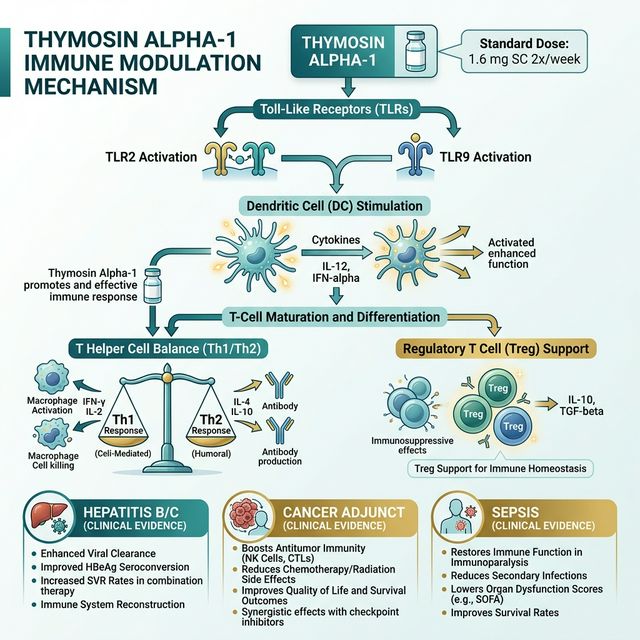
The molecular mechanism: Tα1 acts on toll-like receptors (TLR2, TLR9) on dendritic cells and macrophages, enhancing antigen presentation and the initiation of adaptive immune responses. It promotes T-cell maturation and differentiation — increasing CD4+ T helper cells, CD8+ cytotoxic T cells, and NK cells. It enhances the Th1/Th2 balance, shifting toward Th1 (cell-mediated immunity) which is critical for antiviral and anti-tumor defense. It stimulates production of regulatory T-cells (Tregs) that prevent autoimmune overshoot.
This dual action — enhancing pathogen-directed immunity while supporting immune regulation — is what makes Tα1 unique. In chronic hepatitis B, it enhances viral clearance. In sepsis, it restores immune competence in exhausted immune systems. In cancer, it enhances anti-tumor surveillance. In autoimmune thyroiditis, it modulates the immune response without exacerbating autoimmunity. No other single peptide has demonstrated this breadth of immune modulation.
Clinical Evidence Across Multiple Conditions
(1) Chronic Hepatitis B — multiple randomized controlled trials demonstrate that Tα1 (1.6 mg subcutaneously twice weekly for 24-52 weeks) achieves viral suppression and seroconversion rates comparable to interferon-alpha, with dramatically fewer side effects. This is the indication for which Tα1 is most widely approved globally.
(2) Cancer immunotherapy adjunct — Tα1 has been used alongside chemotherapy and radiation in hepatocellular carcinoma, non-small-cell lung cancer, melanoma, and breast cancer. Meta-analyses show improved overall survival and reduced chemotherapy-related immune suppression when Tα1 is added to standard cancer therapy. The mechanism: Tα1 counteracts the immunosuppressive effects of chemotherapy, maintaining immune surveillance during treatment.
(3) Sepsis and critical illness — in severe sepsis, the immune system often enters a state of "immunoparalysis" where excessive initial inflammation is followed by profound immune suppression. Tα1 has been studied in sepsis ICU patients, showing reduced 28-day mortality in some trials. The Chinese Critical Care Medicine Society includes Tα1 in their sepsis treatment guidelines.
(4) Vaccine adjuvant — Tα1 enhances vaccine responses in immunocompromised populations (elderly, chronic disease patients) who typically mount weak responses to standard vaccination. Studies with influenza and hepatitis B vaccines show improved antibody titers when Tα1 is co-administered.
Dosing, Access, and the Category 2 Obstacle
Standard dosing from clinical trials: 1.6 mg subcutaneously, twice weekly. This is the dose that appears across virtually all major Tα1 clinical trials. Some practitioners use daily dosing (1.6 mg/day) for acute immune challenges or cancer adjunct protocols.
A standard 5 mg vial reconstituted with 1 mL BAC water yields a concentration of 5 mg/mL = 5,000 mcg/mL. For a 1.6 mg (1,600 mcg) dose: draw 32 units on a U-100 syringe. This gives approximately 3 doses per vial. The CalcMyPeptide reconstitution calculator can verify this math for your specific vial size and BAC water volume.
The access challenge: Tα1 was placed on the FDA Category 2 list in November 2023, restricting compounding pharmacy production in the US. This is arguably the most consequential Category 2 designation because Tα1 has the strongest clinical evidence of any Category 2 peptide. As discussed in our Category 2 article, the 2026 review may reinstate Tα1 to Category 1. Until then, access in the US is limited to research peptide vendors or individual patient INDs. In most other countries, Tα1 (Zadaxin) is available by prescription.
