⚕️ Medical Disclaimer
**⚕️ Medical Disclaimer:** This article is for educational and informational purposes only. It does not constitute medical advice. Consult a qualified healthcare provider before using any peptide.
What Is Survodutide and Why Does It Matter?
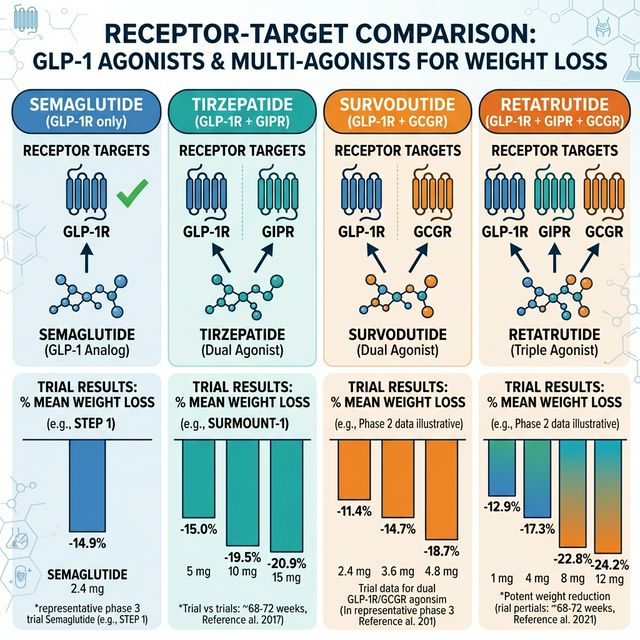
Survodutide (BI 456906) is a dual GLP-1/glucagon receptor agonist developed by Boehringer Ingelheim and Zealand Pharma. While semaglutide targets GLP-1 receptors alone and tirzepatide targets GLP-1 + GIP receptors, survodutide adds glucagon receptor agonism — a fundamentally different approach to weight loss and metabolic improvement.
*Last updated: March 2026*
Here is why that matters: glucagon drives hepatic fat oxidation and energy expenditure in ways that GLP-1 alone cannot. By engaging the glucagon receptor alongside GLP-1, survodutide ramps up thermogenesis and liver fat clearance while simultaneously reducing appetite. The result, in clinical trials, has been weight loss numbers that rival or exceed tirzepatide — with the added benefit of dramatic improvements in liver fat (MASLD/MASH).
This is the drug class that the obesity medicine world is watching most closely in 2026. Let us break down the mechanism, the data, and what it means if you are currently on a GLP-1 protocol. Use our GLP-1 Dose Escalation Guide and Semaglutide Calculator for current protocol tools while survodutide completes its approval pathway.
Mechanism of Action: GLP-1 + Glucagon Dual Agonism
**Survodutide activates two receptors simultaneously: GLP-1R (appetite suppression, insulin secretion, gastric slowing) and GCGR (hepatic fat oxidation, thermogenesis, energy expenditure).** This creates a synergistic metabolic effect that neither pathway achieves alone.
**GLP-1R activation (shared with semaglutide, tirzepatide):**
• Suppresses appetite via hypothalamic signaling
• Slows gastric emptying (increases satiety)
• Enhances glucose-dependent insulin release
• Reduces glucagon secretion from alpha cells (paradoxically, the exogenous glucagon agonism overrides this)
**GCGR activation (unique to survodutide vs semaglutide/tirzepatide):**
• Increases hepatic glycogenolysis and gluconeogenesis (managed by the GLP-1 component's insulin-enhancing effects)
• Drives fatty acid oxidation in the liver — directly burns liver fat
• Increases resting energy expenditure (thermogenesis) by 100-200 kcal/day in preclinical models
• Promotes amino acid catabolism (protein turnover) — important consideration for lean mass preservation
The critical insight: glucagon receptor agonism in isolation would raise blood sugar dangerously. But combined with GLP-1R agonism, the insulin-enhancing effects of GLP-1 counterbalance the hyperglycemic effects of glucagon, creating a net effect of improved glycemia *plus* enhanced fat oxidation. This is pharmaceutical engineering at its most elegant (Ambery et al., 2018, PMID: 30407052).
How Does Survodutide Compare to Semaglutide, Tirzepatide, and Retatrutide?
The incretin/glucagon space is crowded. Here is where survodutide sits:
| Compound | Receptor Targets | Developer | Phase | Max Weight Loss (Trials) | Liver Fat Reduction | Unique Angle |
|---|---|---|---|---|---|---|
| Semaglutide (Wegovy) | GLP-1R | Novo Nordisk | Approved | ~15-17% | Moderate | First mover, widest data |
| Tirzepatide (Zepbound) | GLP-1R + GIPR | Eli Lilly | Approved | ~20-22.5% | Good | Dual incretin, best-in-class approved |
| Survodutide | GLP-1R + GCGR | Boehringer/Zealand | Phase 3 | ~19-20% | Excellent (up to 83%) | Liver-focused fat oxidation |
| Retatrutide | GLP-1R + GIPR + GCGR | Eli Lilly | Phase 3 | ~24% | Very good | Triple agonist, highest efficacy |
| Orforglipron | GLP-1R (non-peptide) | Eli Lilly | Phase 3 | ~14.7% | Moderate | Oral small molecule |
Survodutide's key differentiator is its dramatic effect on liver fat. In Phase 2 trials (SYNCHRONIZE-1), survodutide reduced liver fat content by up to 83%, compared to 47-53% for semaglutide in comparable endpoints (Sanyal et al., 2024, PMID: 38587249). This makes it a frontrunner for MASLD/MASH treatment.
For more on how orforglipron (the oral alternative) fits in, read our Orforglipron guide. For context on retatrutide, check our retatrutide deep dive.
Clinical Trial Data: Phase 2 and Phase 3 Results
**Phase 2 (SYNCHRONIZE-1, published 2024):** 334 adults with BMI ≥27 were randomized to survodutide (0.6-4.8 mg weekly) or placebo for 46 weeks. Results at the 4.8 mg dose:
• Mean body weight reduction: **-18.7%** (vs -2.1% placebo)
• ≥5% weight loss: 96.2% of participants
• ≥15% weight loss: 63.5% of participants
• Liver fat reduction: up to **-83%** in the subset with elevated baseline liver fat
• HbA1c improvement: significant in subjects with prediabetes
**Phase 3 (SYNCHRONIZE program, 2025-2026):** The Phase 3 program includes multiple trials:
• **SYNCHRONIZE-2:** Survodutide vs placebo in adults with obesity (primary outcome: % body weight change at 72 weeks)
• **SYNCHRONIZE-3:** Survodutide in adults with obesity and type 2 diabetes
• **SYNCHRONIZE-NASH:** Survodutide for MASH (metabolic dysfunction-associated steatohepatitis) with liver fibrosis
If Phase 3 results replicate Phase 2, survodutide will likely receive FDA submission in late 2026 or early 2027. The MASH indication may arrive first, given the unmet medical need and FDA's prioritization pathway.
Make sure your blood work panels include liver function tests if you are monitoring liver-related peptide effects — ALT, AST, and ideally a FibroScan or MRI-PDFF for liver fat quantification.
Side Effects and Tolerability Profile
**Survodutide's side effect profile is consistent with the GLP-1 class — primarily GI — but the glucagon component adds unique considerations:**
• **Nausea, vomiting, diarrhea:** Most common, occurring in 30-50% of participants at higher doses. Dose escalation mitigates this — a gradual titration schedule (similar to semaglutide escalation) is essential.
• **Decreased appetite:** Expected and therapeutic. This is the mechanism of action.
• **Heart rate increase:** Small but statistically significant (+2-5 bpm). Monitor if you have cardiovascular risk factors.
• **Potential hyperglycemia risk:** The glucagon component could theoretically raise blood sugar in susceptible individuals, though the GLP-1 component counterbalances this. Regular HbA1c and fasting glucose monitoring is critical. See our lab guide for the full panel recommendation.
• **Lean mass loss:** Like all drugs causing rapid weight loss, survodutide carries lean mass preservation concerns. Resistance training and adequate protein intake (1.6-2.2 g/kg/day) are non-negotiable during any significant weight loss intervention.
For a complete safety comparison across peptide classes, review our peptide safety guide and drug interactions guide.
What Does Survodutide Mean for Current GLP-1 Users?
If you are currently on semaglutide or tirzepatide and wondering about survodutide, here is the practical take:
• **Survodutide is not yet available.** It remains in Phase 3 trials. Switching is not currently possible outside of clinical trial enrollment.
• **If approved, it will likely be a second-line or liver-targeted option.** Semaglutide and tirzepatide already have massive real-world safety datasets. Survodutide will carve its niche for patients with liver fat accumulation or those who have plateaued on existing GLP-1s.
• **The glucagon component makes it particularly interesting for MASLD/MASH patients** — a population poorly served by current approved therapies.
• **For compounding pharmacies:** Survodutide is a dual-agonist peptide, not a small molecule. Compounding feasibility will depend on patent expiry and regulatory status. Use our Semaglutide Calculator for current GLP-1 dosing while the pipeline matures.
• **Track your high-dose oral semaglutide options** for the latest on formulation advances in the meantime.
Frequently Asked Questions
**When will survodutide be FDA-approved?** Based on the Phase 3 timeline, FDA submission is expected in late 2026 or 2027. Approval, if granted, would likely follow 10-12 months after submission. The MASH indication may be prioritized.
**Is survodutide better than semaglutide for weight loss?** Phase 2 data shows comparable weight loss (~18.7% vs ~15-17% for semaglutide at maximum dose) but dramatically superior liver fat reduction (83% vs ~47%). Head-to-head trials are needed for definitive comparison.
**Can survodutide cause hypoglycemia?** In clinical trials, hypoglycemia rates were low — GLP-1R agonism provides glucose-dependent insulin secretion, meaning insulin is primarily released when glucose is elevated. The glucagon component could theoretically raise glucose, but the dual mechanism appears to be self-balancing in trials.
**How does survodutide differ from retatrutide?** Retatrutide is a triple agonist (GLP-1R + GIPR + GCGR) while survodutide is a dual agonist (GLP-1R + GCGR). Retatrutide adds GIP receptor engagement, which provides additional insulin-tropic benefit. Both include glucagon agonism. Retatrutide has shown higher peak weight loss (~24% in Phase 2) but direct comparison awaits Phase 3.
**Should I get specific lab work if considering survodutide in the future?** Yes — start baseline labs now, including liver function (ALT, AST, GGT), liver fat assessment (if available via imaging), HbA1c, and a lipid panel. Use our blood work guide for the complete checklist.
Final Word
**⚕️ Medical Disclaimer:** This article is for educational and informational purposes only. It does not constitute medical advice. Consult a qualified healthcare provider before using any peptide.