⚕️ Medical Disclaimer
**⚕️ Medical Disclaimer:** This article is for educational and informational purposes only. It does not constitute medical advice. Consult a qualified healthcare provider before using any compound.
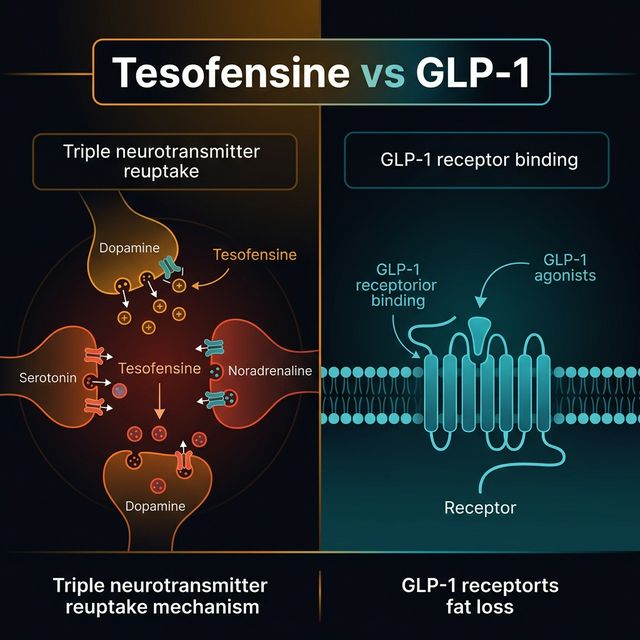
What Is Tesofensine and How Does It Cause Weight Loss?
Tesofensine is a triple monoamine reuptake inhibitor (serotonin, dopamine, norepinephrine) that produces significant weight loss through appetite suppression and increased energy expenditure. Originally developed by NeuroSearch for Parkinson's and Alzheimer's disease, weight loss was discovered as a prominent "side effect" in Phase 2 trials. The TIPO-1 trial showed 10.6% mean weight loss at 0.5 mg in 24 weeks — more than any anti-obesity drug approved at the time (Astrup et al., 2008, PMID: 18978768).
*Last updated: March 2026*
Tesofensine Dosing Protocol
**Standard dose:** 0.25-0.5 mg orally, once daily. **Starting dose:** 0.25 mg for the first 2 weeks, then increase to 0.5 mg. **Form:** oral capsule — no reconstitution or injection required. **Cycle:** typically 12-24 weeks. Not FDA-approved; available through compounding pharmacies in some jurisdictions.
Because tesofensine is an oral compound (not a peptide), it does not require reconstitution. However, for users stacking with injectable peptides, use the Reconstitution Calculator for those vials.
Tesofensine vs Semaglutide vs Tirzepatide: Weight Loss Comparison
| Feature | Tesofensine | Semaglutide (Wegovy) | Tirzepatide (Mounjaro) |
|---|---|---|---|
| Mechanism | Triple monoamine reuptake inhibitor | GLP-1R agonist | GLP-1R + GIPR dual agonist |
| Weight Loss | ~10.6% (24 wk) | ~14.9% (68 wk) | ~22.5% (72 wk) |
| Route | Oral capsule | SubQ weekly injection | SubQ weekly injection |
| Heart Rate | +7.4 bpm increase | Slight increase | Slight increase |
| GI Side Effects | Dry mouth, constipation | Nausea (44%), vomiting (24%) | Nausea (31%), diarrhea (21%) |
| FDA Status | Not approved | Approved (2021) | Approved (2023, obesity) |
| Muscle Preservation | Better (dopamine-mediated) | Poor (25-40% lean mass loss) | Better than sema |
For GLP-1 dosing, see our semaglutide dose calculator and GLP-1 escalation guide.
Can Tesofensine Be Stacked with GLP-1 Peptides?
Yes — tesofensine + low-dose semaglutide is an emerging stack in the compounding pharmacy space. The rationale: tesofensine suppresses appetite via monoamine pathways (dopamine/serotonin) while semaglutide works through GLP-1R/satiety signaling. Different receptor targets, potentially additive effects. However, combination use requires careful cardiovascular monitoring (both can elevate heart rate). See our stacking 101 for general principles.
Side Effects and Cardiovascular Considerations
Primary concerns: **heart rate increase** (+7.4 bpm at 0.5 mg) and **blood pressure elevation** (modest) — these cardiovascular effects are the main reason tesofensine hasn't received FDA approval despite strong weight loss efficacy. Other side effects: dry mouth (most common), constipation, insomnia, and mild mood elevation. No cases of pulmonary hypertension (the issue that sank fenfluramine/dexfenfluramine).
Contraindicated in uncontrolled hypertension, cardiovascular disease, history of stroke, and concurrent use of MAOIs. See our peptide side effects guide for broader context.
Frequently Asked Questions
**Is tesofensine a peptide?** No — it is a small molecule (not a peptide). We cover it because the peptide community frequently uses it alongside GLP-1 peptides for fat loss.
**Does tesofensine improve mood?** The dopamine reuptake inhibition can produce mild mood elevation and increased motivation — an effect distinct from GLP-1 peptides, which some users report cause emotional blunting.
**Will tesofensine ever be FDA-approved?** The Mexican pharmaceutical company Sanfer completed Phase 3 trials (TENOR study) and obtained Mexican regulatory approval. US FDA approval remains uncertain due to cardiovascular signal concerns.
**Can I use tesofensine with intermittent fasting?** Yes — as an oral capsule, take it in the morning with or without food. It synergizes well with fasting protocols. See peptides + IF.